Inflammation and Blood Sugar: How CRP and Glucose Interact
Chronic inflammation and blood sugar exist in a bidirectional cycle. hs-CRP risk tiers, the NF-kB pathway, oxidative stress from glucose spikes, IL-6, TNF-alpha, and anti-inflammatory dietary strategies.
The Bidirectional Relationship Between Inflammation and Blood Sugar
Chronic low-grade inflammation and elevated blood sugar form a self-reinforcing cycle in which each condition worsens the other. High blood glucose — particularly postmeal spikes above 140 mg/dL — triggers oxidative stress by overwhelming the mitochondrial electron transport chain, producing excess reactive oxygen species (ROS) that damage endothelial cells lining blood vessels. This oxidative damage activates the nuclear factor kappa-B (NF-kB) signaling pathway, the master regulator of inflammatory gene expression, which upregulates production of pro-inflammatory cytokines including interleukin-6 (IL-6), tumor necrosis factor-alpha (TNF-alpha), and C-reactive protein (CRP). These inflammatory mediators, in turn, impair insulin signaling by disrupting insulin receptor substrate-1 (IRS-1) phosphorylation — the critical first step in the insulin signaling cascade inside cells. The result is worsened insulin resistance, which produces higher glucose spikes, which triggers more inflammation. A 2016 study in Nature Reviews Immunology described this as the "metabolic inflammation" paradigm and estimated that chronic low-grade inflammation accounts for 30-40% of the insulin resistance observed in obesity. Understanding this cycle is essential because breaking it at either point — reducing glucose spikes or reducing inflammation — produces improvements in both directions simultaneously.
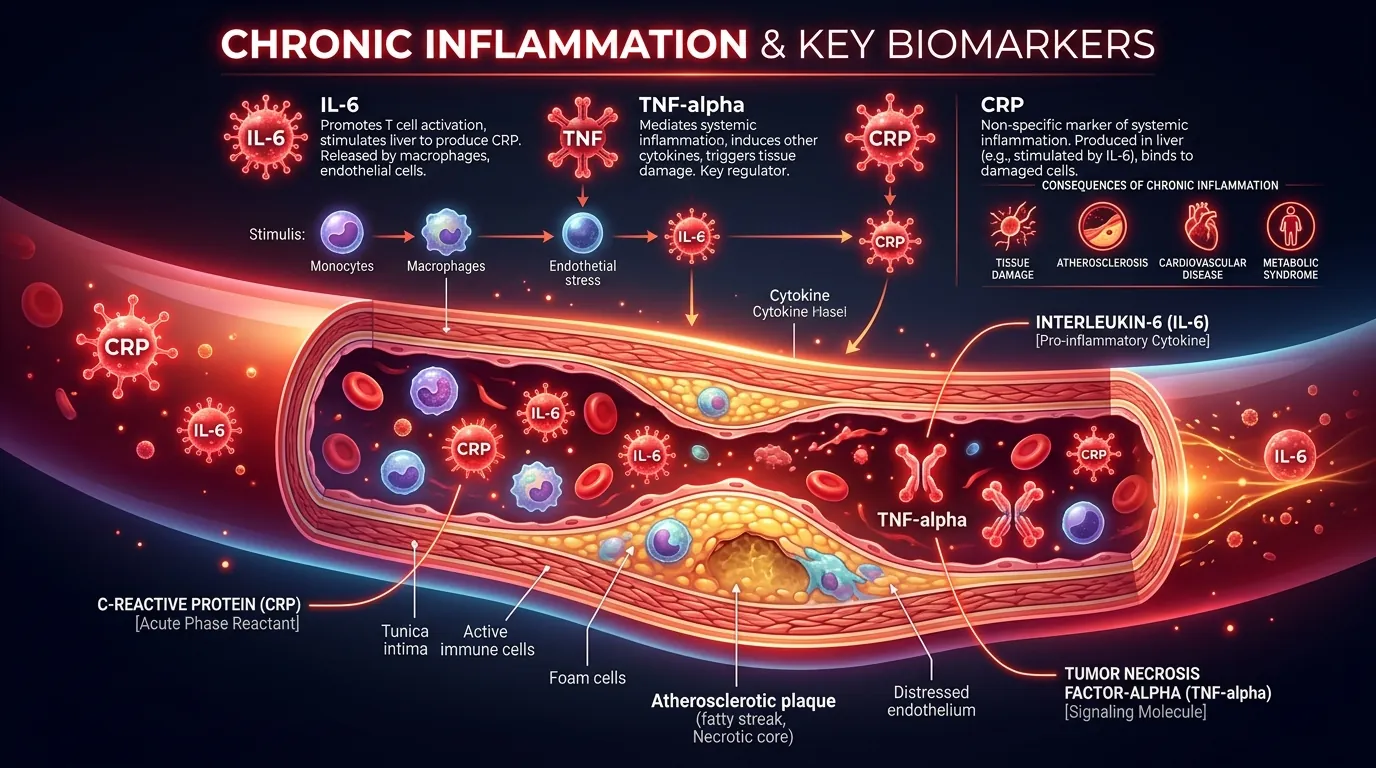
hs-CRP: The Primary Inflammation Marker for Metabolic Health
High-sensitivity C-reactive protein (hs-CRP) is the most widely validated blood marker for systemic inflammation and cardiovascular risk. CRP is an acute-phase protein produced by the liver in response to IL-6 signaling, and its blood concentration rises proportionally to the degree of systemic inflammation. The "high-sensitivity" assay detects CRP at concentrations below 10 mg/L — the threshold for standard CRP tests used to detect acute infections — enabling quantification of the low-grade chronic inflammation associated with metabolic disease. The American Heart Association and CDC define 3 cardiovascular risk tiers based on hs-CRP: low risk at below 1.0 mg/L, moderate risk at 1.0-3.0 mg/L, and high risk at above 3.0 mg/L. A 2002 New England Journal of Medicine study (Ridker et al.) demonstrated that hs-CRP predicted cardiovascular events independently of LDL cholesterol — individuals with low LDL but high hs-CRP had higher event rates than those with high LDL but low hs-CRP. The landmark JUPITER trial (2008) further proved that statin therapy reduced cardiovascular events by 44% in patients with normal LDL but elevated hs-CRP (above 2.0 mg/L), establishing hs-CRP as an actionable biomarker, not merely a risk indicator. For metabolic health assessment, hs-CRP provides a quantitative measure of the inflammatory burden that drives insulin resistance and should be included in any comprehensive metabolic panel.
How Glucose Spikes Drive Oxidative Stress and Inflammation
Postmeal glucose spikes — particularly those exceeding 140 mg/dL — produce acute bursts of oxidative stress that accumulate into chronic vascular damage over months and years. The mechanism is well characterized: excess glucose entering cells overwhelms the mitochondrial electron transport chain, causing electrons to "leak" and react with oxygen to form superoxide radicals. Superoxide activates 4 damaging pathways simultaneously: (1) the polyol pathway, which depletes NADPH and reduces the cell's antioxidant capacity; (2) advanced glycation end-products (AGEs), which cross-link proteins and damage structural components of blood vessel walls; (3) the protein kinase C (PKC) pathway, which increases vascular permeability and promotes blood clotting; and (4) the hexosamine pathway, which alters gene expression to favor inflammation. A 2012 study in Diabetes Care by Monnier et al. demonstrated that glucose variability — the magnitude and frequency of glucose swings throughout the day — produced more oxidative stress markers than sustained high glucose of the same average level. This means a person with an average glucose of 120 mg/dL who swings between 80 and 200 mg/dL experiences more oxidative damage than someone with a steady glucose of 140 mg/dL. CGM data showing high coefficient of variation (above 36%) is therefore a direct indicator of elevated oxidative stress and inflammatory risk.
The NF-kB Pathway: How Inflammation Becomes Insulin Resistance
Nuclear factor kappa-B (NF-kB) is a protein complex that functions as the master switch for inflammatory gene expression in nearly all human cells. In metabolically healthy tissue, NF-kB remains inactive in the cytoplasm, bound to an inhibitory protein called IkB. When oxidative stress, TNF-alpha, or other danger signals reach the cell, IkB kinase (IKK) phosphorylates and degrades IkB, freeing NF-kB to enter the nucleus and activate transcription of over 150 inflammatory genes. In the context of metabolic disease, NF-kB activation in liver, fat, and muscle tissue produces 3 consequences that directly worsen insulin resistance: (1) increased production of TNF-alpha, which phosphorylates IRS-1 at serine residues instead of tyrosine residues — a critical modification that blocks insulin signal transduction; (2) increased IL-6 production, which activates the SOCS-3 pathway that degrades insulin receptors on cell surfaces; and (3) increased production of MCP-1 (monocyte chemoattractant protein-1), which recruits macrophages into adipose tissue where they sustain chronic inflammation. A 2013 study in Cell Metabolism demonstrated that selectively blocking NF-kB in liver cells improved insulin sensitivity by 45% in obese mice, confirming that NF-kB-driven inflammation is a direct cause — not merely a consequence — of insulin resistance.
Anti-Inflammatory Dietary Strategies for Metabolic Health
Dietary modification is the most accessible intervention for reducing the chronic inflammation that drives metabolic dysfunction. The Mediterranean dietary pattern — characterized by olive oil, fatty fish, vegetables, nuts, legumes, and limited red meat and refined carbohydrates — reduced hs-CRP by 20% and IL-6 by 15% over 12 months in the PREDIMED trial (N = 7,447). Specific anti-inflammatory foods supported by clinical evidence include: fatty fish (salmon, sardines, mackerel) providing 2-4 grams of EPA and DHA omega-3 fatty acids per week, which inhibit NF-kB activation and reduce CRP by 20-35% according to a 2019 meta-analysis in Atherosclerosis (N = 4,973); extra-virgin olive oil containing oleocanthal, a compound that inhibits COX-2 with potency comparable to low-dose ibuprofen; berries (blueberries, strawberries) rich in anthocyanins that scavenge superoxide radicals; turmeric (curcumin) at 500-1000 mg/day, which inhibits NF-kB and reduced CRP by 36% in a 2019 meta-analysis of 11 trials; and green tea (EGCG), which inhibits TNF-alpha at doses of 3-5 cups daily. Conversely, pro-inflammatory foods that should be limited include refined sugar (activates NF-kB within 2 hours of consumption), trans fats (directly impair endothelial function), processed meats (contain AGE precursors), and excessive omega-6 vegetable oils (shift the omega-6 to omega-3 ratio toward inflammation). CGM data can guide anti-inflammatory eating by identifying which specific meals produce the largest glucose spikes — and therefore the most oxidative stress and inflammatory activation — allowing individuals to make targeted substitutions rather than following generic dietary rules.