CGM Sensor Technology: Electrochemical, Fluorescence, and Biosensor Systems
How CGM sensors measure glucose — electrochemical glucose oxidase, fluorescence-based Eversense, and emerging biosensor technologies compared for 2026.
How CGM Sensors Detect Glucose
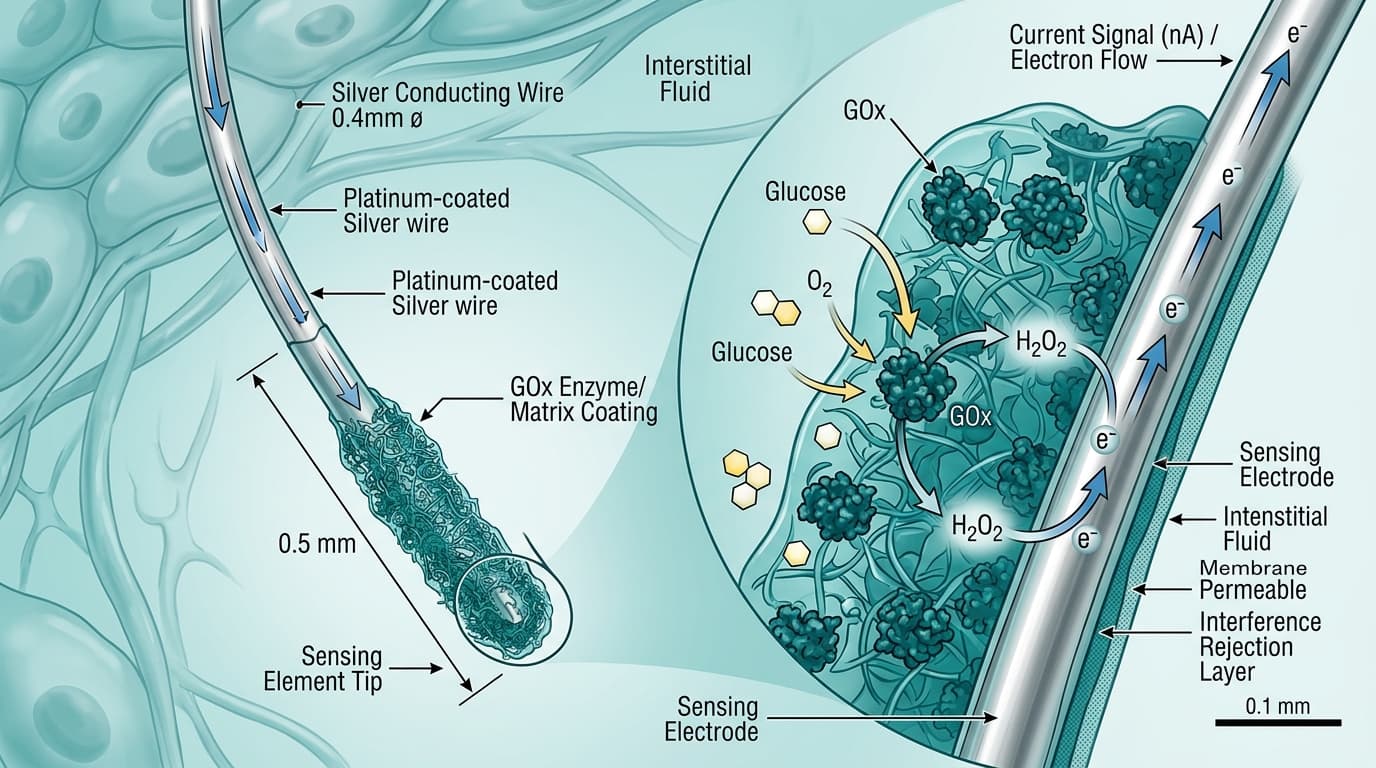
Every continuous glucose monitor relies on a biosensor that converts glucose concentration into a measurable signal. The sensor is a thin, flexible filament — typically 5 to 7 millimeters long and 0.35 millimeters in diameter — inserted into the subcutaneous tissue just beneath the skin. Once in place, the filament is surrounded by interstitial fluid (ISF), the liquid that bathes every cell in your body. Glucose molecules in the ISF interact with the sensor's active surface, triggering a chemical or optical reaction that the transmitter converts into a digital glucose reading. The entire measurement cycle repeats every 1 to 5 minutes, generating 288 to 1,440 data points per day. Two fundamentally different sensing technologies are used in commercial CGMs: electrochemical (enzyme-based) and fluorescence (optical). The choice of technology directly affects sensor lifespan, accuracy stability, and the physical form factor of the device.
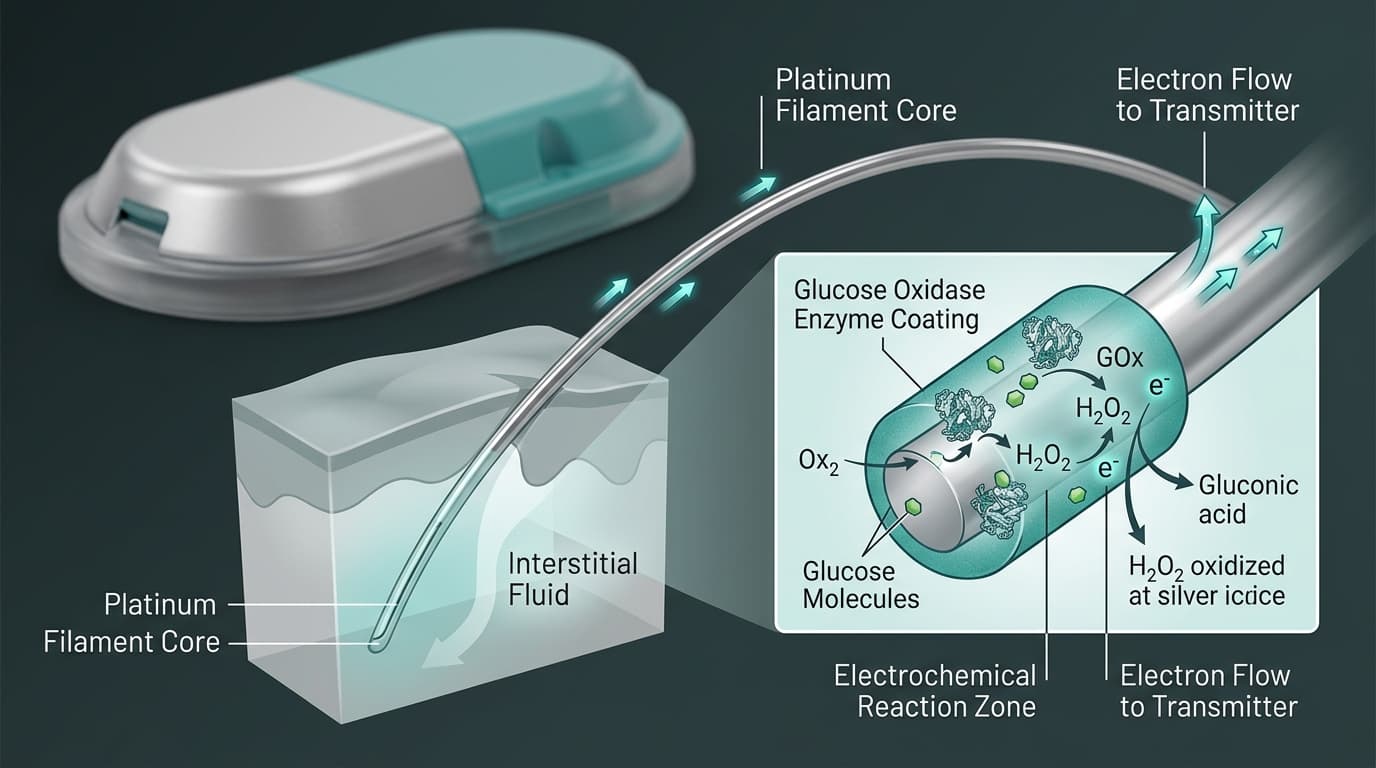
Electrochemical Glucose Oxidase Sensors
The electrochemical approach is the most widely used CGM sensor technology, employed by Dexcom, Abbott, and Medtronic across all of their product lines. The filament is coated with glucose oxidase (GOx), an enzyme that catalyzes the oxidation of glucose into gluconolactone and hydrogen peroxide. The hydrogen peroxide is then oxidized at a working electrode, generating an electrical current proportional to the glucose concentration. A higher glucose level produces a stronger current. The transmitter's analog-to-digital converter reads this current, and a proprietary algorithm translates it into a glucose value in mg/dL. Modern electrochemical sensors achieve a MARD of 7.9% to 9.1% and last 7 to 15 days before the enzyme layer degrades and the sensor must be replaced. One limitation of this approach is that the enzyme is consumed over time, contributing to gradual signal drift that can reduce accuracy toward the end of the sensor's wear period.
Fluorescence-Based Optical Sensors
Senseonics pioneered the fluorescence approach in its Eversense product line. Instead of an enzyme-coated filament, the Eversense sensor is a small cylindrical implant (3.5 mm x 18.3 mm) placed under the skin by a healthcare provider in a brief office procedure. The implant contains a glucose-indicating polymer that changes its fluorescent emission intensity in proportion to the surrounding glucose concentration. An external transmitter worn over the implant site contains an LED that illuminates the sensor through the skin and a photodetector that reads the returning fluorescence signal. Because the fluorescence reaction does not consume glucose or degrade the polymer, the sensor lasts dramatically longer — 180 days for the Eversense E3 and up to 365 days for the Eversense 365. The trade-off is a slightly higher MARD of 8.5% to 9.1% and the requirement for a minor surgical insertion and removal procedure.
Emerging Biosensor Technologies
Several next-generation CGM technologies are in clinical development. Microneedle patch sensors use arrays of painless, dissolvable microneedles to access interstitial fluid without a traditional inserter. Osmotic pressure sensors measure glucose-induced changes in fluid pressure across a semipermeable membrane. Non-invasive optical sensors aim to measure glucose through the skin using near-infrared spectroscopy or Raman scattering, though no non-invasive CGM has achieved the accuracy needed for FDA clearance as of 2026. Biocompatible hydrogel coatings are also being developed to extend sensor life by reducing the foreign body response that causes signal degradation in current electrochemical sensors. These technologies are 3 to 7 years from commercial availability, but they represent the future of continuous glucose monitoring.
How Sensor Construction Affects Performance
The physical construction of a CGM sensor determines its accuracy, comfort, and durability. Thinner filaments (under 0.3 mm) cause less tissue trauma during insertion and reduce the inflammatory response that can skew glucose readings in the first 24 hours. Multi-layer membrane coatings control the rate at which glucose diffuses to the enzyme surface, preventing sensor saturation at high glucose levels and improving linearity across the full measurement range of 40 to 500 mg/dL. Biocompatible outer layers — often polyurethane or silicone — reduce the body's immune response to the foreign material. Electrode design also matters: triple-electrode configurations (working, reference, and counter electrodes) provide more stable measurements than dual-electrode designs by compensating for baseline drift and interference from other molecules like acetaminophen. All of these engineering decisions influence the overall accuracy as measured by MARD.