
CGM Calibration: Factory-Calibrated vs Fingerstick-Based Systems
Modern CGMs use factory calibration — no fingerstick required. How calibration works, which devices still need it, and why factory-calibrated sensors dominate in 2026.
What Is CGM Calibration?
Calibration is the process of aligning a CGM sensor's glucose readings with a known reference value — typically a fingerstick blood glucose measurement. In older CGM systems, the user would perform a fingerstick test, enter the blood glucose value into the CGM app or receiver, and the device's algorithm would adjust its readings to match. This was necessary because raw sensor signals drift over time due to changes in the enzyme layer, tissue inflammation, and variations in interstitial fluid composition. Calibration corrected for this drift and kept readings accurate. First-generation CGMs required 2 to 4 fingerstick calibrations per day, and a missed or inaccurate calibration could cause the CGM to display incorrect readings for hours until the next calibration point.
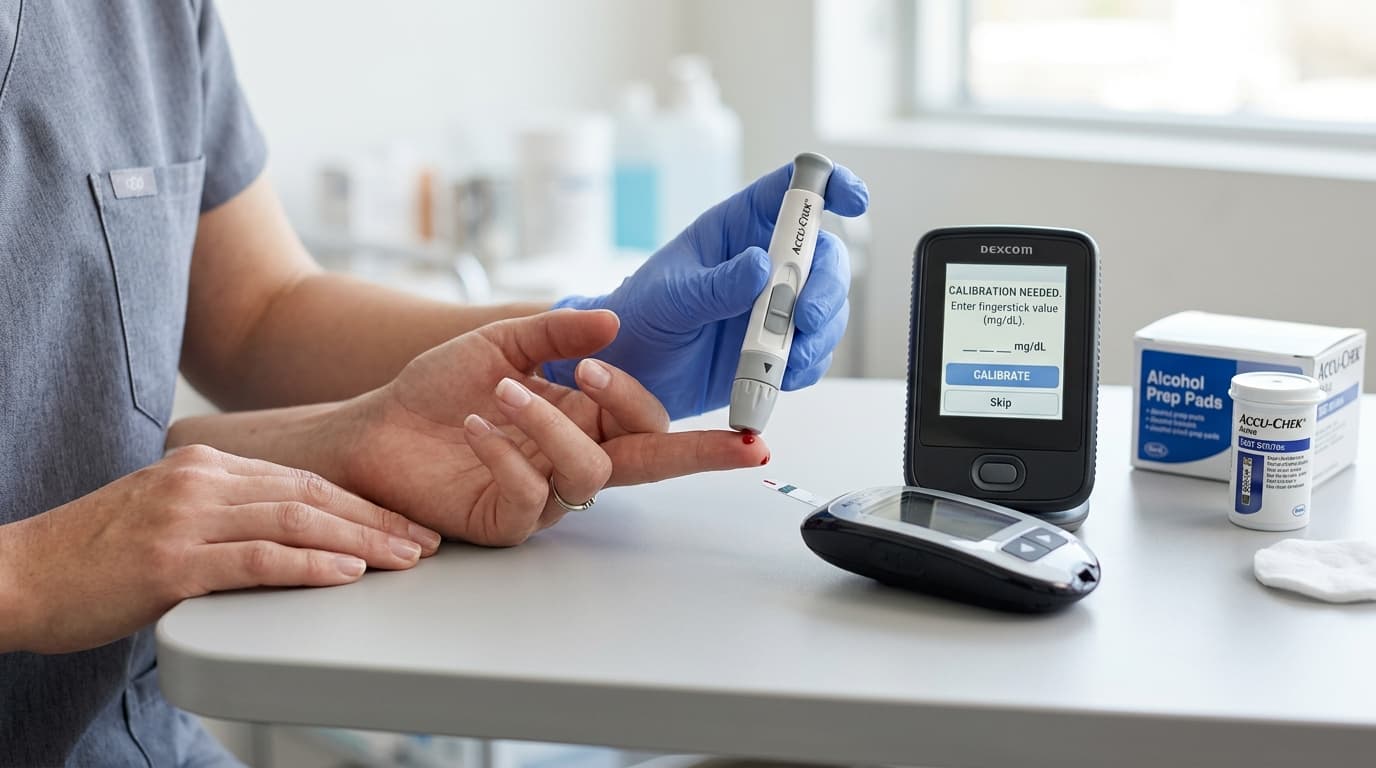
Factory Calibration: How Modern CGMs Eliminate Fingersticks
Factory calibration is the breakthrough that transformed CGMs from high-maintenance devices into consumer-friendly wearables. During manufacturing, each sensor undergoes automated testing against known glucose concentrations. The resulting calibration data — which maps the sensor's raw electrical signal to glucose values across the full measurement range — is encoded into the sensor or its packaging. When the user activates the sensor, the device loads this factory calibration profile and applies it to every reading for the sensor's entire wear period. No fingerstick input is needed. The Dexcom G7, FreeStyle Libre 3, FreeStyle Libre 3 Plus, Dexcom Stelo, and Abbott Lingo are all factory-calibrated and require zero user calibrations. The Eversense E3 is the only current-generation CGM that still requires 1 fingerstick calibration per day during the first 21 days of a new sensor, after which it transitions to factory-calibrated operation for the remainder of its 180-day life.
Does Calibration Affect Accuracy?
Incorrect calibration was historically the leading cause of inaccurate CGM readings. A fingerstick meter that was off by 15% (within FDA tolerance) would propagate that error into the CGM's algorithm, causing all subsequent readings to skew until the next calibration. Calibrating during rapid glucose changes amplified the error because the blood-to-interstitial lag meant the fingerstick and CGM were measuring glucose at different points in time. Factory calibration eliminates this user-dependent error source entirely. Clinical data shows that factory-calibrated devices achieve equivalent or superior MARD compared to user-calibrated predecessors. The Dexcom G6 (factory-calibrated, optional fingerstick calibration) achieved 9.0% MARD, while its predecessor the Dexcom G5 (2 daily calibrations required) achieved 9.0% as well — demonstrating that factory calibration matched human-calibrated accuracy while removing the burden and risk of user error.
When You Should Still Use a Fingerstick Meter
Even with factory-calibrated CGMs, fingerstick blood glucose meters remain important backup tools. The FDA recommends confirming CGM readings with a fingerstick when the CGM reading does not match your symptoms (for example, the CGM shows 120 mg/dL but you feel low), during the first 24 hours after inserting a new sensor when readings may be less stable, when glucose is changing rapidly (rising or falling faster than 3 mg/dL per minute), and if the CGM displays a sensor error or data gap. People on intensive insulin therapy should always have a fingerstick meter accessible. For non-diabetic wellness users of over-the-counter CGMs like the Dexcom Stelo, fingerstick confirmation is rarely necessary since readings are used for trend awareness rather than treatment decisions.