How Continuous Glucose Monitors Work: 9 Core Technologies Explained
A continuous glucose monitor is a wearable device that measures blood sugar levels every 1 to 5 minutes through a tiny sensor inserted just beneath the skin. This page explains every layer of CGM technology — from the electrochemical reaction at the sensor tip to the Bluetooth signal that delivers a reading to your smartphone.
What Is a CGM and Why Does It Matter?
A continuous glucose monitor (CGM) replaces traditional fingerstick blood glucose testing with an automated, around-the-clock measurement system. Instead of pricking your finger 4 to 10 times per day and receiving a single snapshot, a CGM captures 288 or more glucose readings every 24 hours and displays them as a continuous trend line. This density of data reveals patterns — postmeal spikes, overnight lows, exercise effects, and the impact of stress — that isolated fingerstick readings cannot detect.
The technology traces its origins to subcutaneous glucose sensing research in the 1960s, but consumer-ready CGMs only became practical after Medtronic launched the first commercial system in 1999. Since then, three generations of sensors have reduced size, improved accuracy, and eliminated the need for routine fingerstick calibration. Today, over 4.5 million people in the United States use a CGM, and the FDA has approved over-the-counter models for adults without diabetes who want to track their metabolic health. For a ranked comparison of every device currently available, see the best continuous glucose monitors in 2026.
CGM Sensor Types: Electrochemical vs Fluorescence
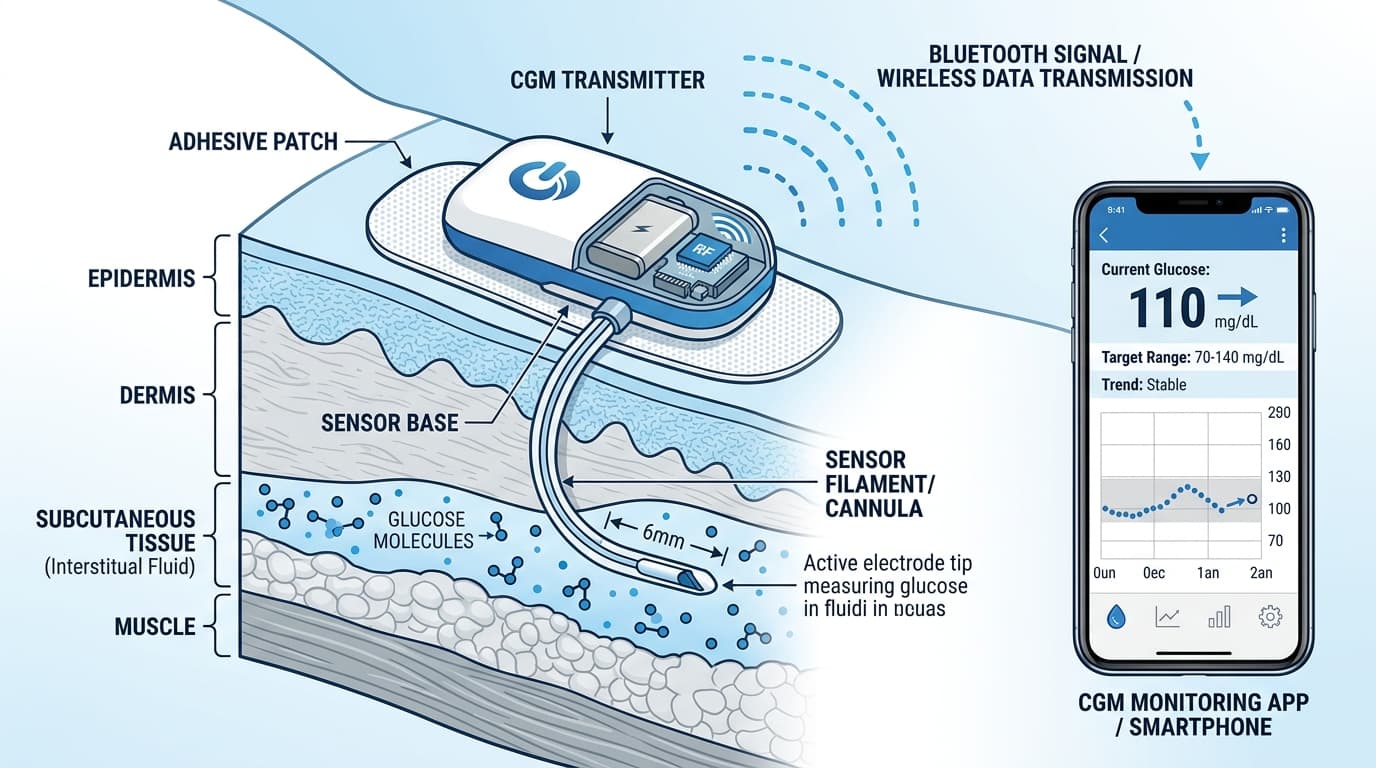
Every CGM starts with a biosensor — a small filament or implant that interacts with glucose molecules in the interstitial fluid (the liquid between your cells). Two fundamentally different sensing technologies exist on the market today.
Electrochemical Sensors
Used by Dexcom, Abbott, and Medtronic, electrochemical sensors coat a thin filament with glucose oxidase enzyme. When glucose contacts the enzyme, a chemical reaction produces hydrogen peroxide, which generates a measurable electrical current. The current is proportional to the glucose concentration. These sensors are disposable, worn for 7 to 15 days, and achieve a MARD as low as 7.9%.
Fluorescence Sensors
Used exclusively by Senseonics in the Eversense product line, fluorescence sensors contain a glucose-sensitive polymer that changes its light emission when exposed to glucose. An LED in the external transmitter illuminates the implanted sensor, and a photodetector reads the resulting fluorescence intensity. Because the sensor does not consume glucose during measurement, it lasts significantly longer — up to 365 days for the Eversense 365.
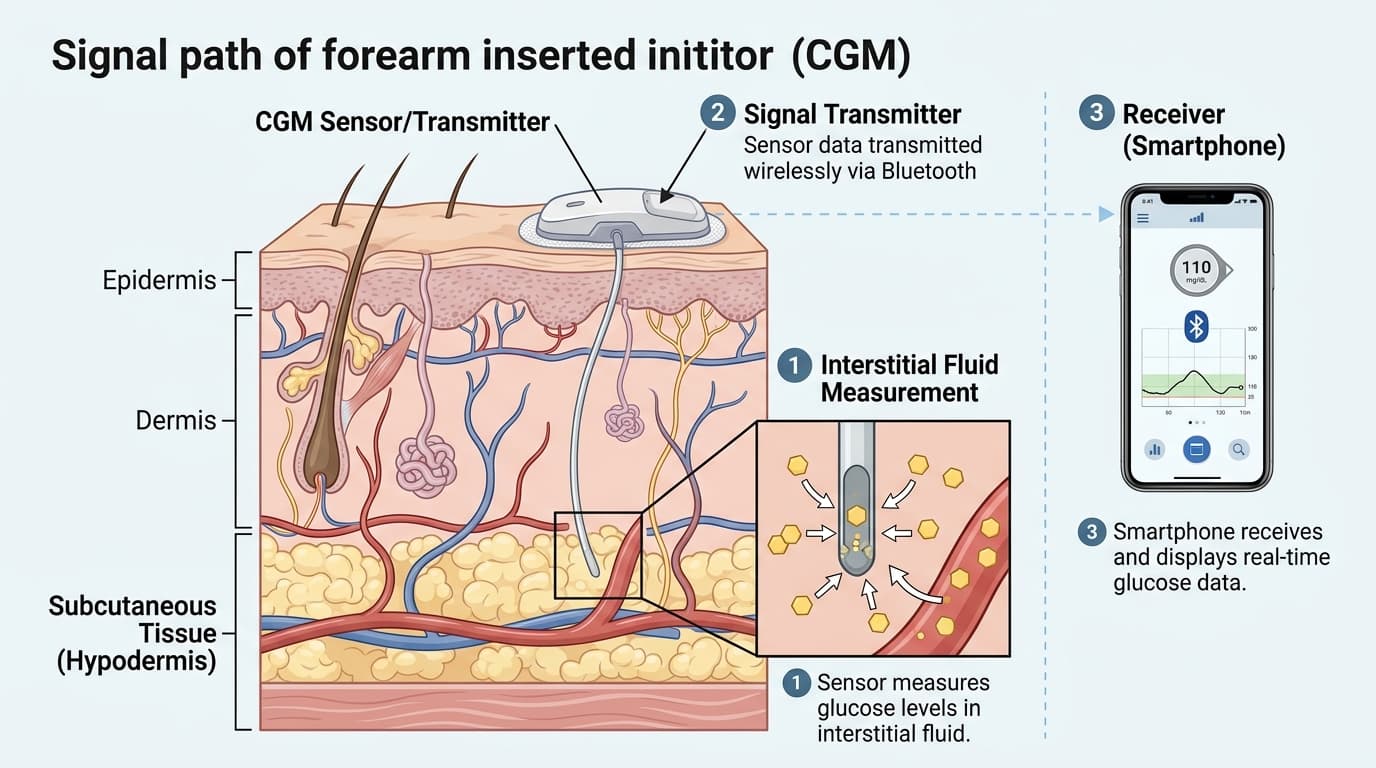
How CGM Readings Are Transmitted
Raw sensor data must travel from the subcutaneous filament to a device you can read — typically a smartphone. The journey involves 3 stages. First, the sensor generates an analog signal (electrical current or fluorescence intensity). Second, the on-body transmitter converts that analog signal into a digital glucose value using a proprietary algorithm. Third, the transmitter broadcasts the digital reading via Bluetooth Low Energy (BLE) to a paired smartphone, smartwatch, or dedicated receiver. The entire process takes less than 5 seconds and repeats every 1 to 5 minutes depending on the device. The Dexcom G7 sends readings every 5 minutes. The FreeStyle Libre 3 reports every minute. Transmitter range is typically 6 to 10 meters (20 to 33 feet), allowing the phone to be in another room while data flows continuously.
Once the reading reaches the smartphone app, it joins a time-series database that can be exported as a glucose report or shared with healthcare providers through cloud platforms like Dexcom Clarity, LibreView, and Glooko.
CGM Accuracy: Understanding MARD
Mean Absolute Relative Difference (MARD) is the gold standard metric for measuring how accurately a CGM sensor tracks true blood glucose. It represents the average percentage difference between CGM readings and simultaneous laboratory-grade blood glucose measurements. A MARD of 8% means, on average, the CGM reading is within 8% of the actual blood glucose value. Lower MARD equals higher accuracy. The best CGMs in 2026 achieve a MARD between 7.9% and 9.1%, which is comparable to the accuracy of consumer-grade fingerstick meters (FDA-allowed range: plus or minus 15%).
Accuracy varies throughout the sensor's life. Readings tend to be less accurate during the first 12 to 24 hours (warmup stabilization) and during rapid glucose changes exceeding 2 mg/dL per minute. For a deep dive into MARD calculations and device-by-device comparisons, see CGM Accuracy and MARD Explained and the MARD glossary entry.
The 3 Generations of Continuous Glucose Monitors
Generation 1: Retrospective CGM (1999-2008)
The earliest CGMs recorded glucose data to an internal memory chip and required a clinic visit to download the information. Users could not see their glucose in real time. Accuracy was poor (MARD 15-20%), sensors lasted 3 days, and 4 daily fingerstick calibrations were mandatory. These devices served primarily as diagnostic tools, not daily management devices.
Generation 2: Real-Time CGM with Calibration (2008-2018)
Devices like the Dexcom G4, G5, and Medtronic Guardian connected to dedicated receivers and displayed real-time glucose values. Accuracy improved to MARD 9-12%, and sensors lasted 7 days. However, users still needed 2 fingerstick calibrations per day. These systems introduced high/low glucose alarms and insulin pump integration for the first time.
Generation 3: Factory-Calibrated CGM (2018-Present)
Current-generation devices — the Dexcom G7, FreeStyle Libre 3, and Eversense E3 — are factory-calibrated at the point of manufacture, eliminating routine fingerstick calibrations for most users. MARD has reached 7.9-9.1%, sensor wear times extend to 10-15 days (or 365 days for Eversense 365), and Bluetooth connectivity sends data directly to smartphones. The FDA has also created an over-the-counter pathway, enabling devices like the Dexcom Stelo and Abbott Lingo to be sold without a prescription.
CGM Accuracy: How Reliable Is the Data?
Every CGM measures glucose in interstitial fluid (ISF) — the thin layer of fluid surrounding cells beneath the skin — not directly in blood. Interstitial fluid is a plasma-derived liquid that fills the spaces between cells in all body tissues, and its glucose concentration closely tracks blood glucose but with a time delay. This creates a 5-15 minute physiological lag between blood glucose changes and CGM readings. During rapid glucose rises (after eating a high-glycemic meal) or drops (during vigorous exercise), CGM readings trail actual blood glucose by this lag time. The lag is a physical limitation of where the sensor sits in the body, not a technology deficiency — even a theoretically perfect sensor in interstitial fluid would exhibit this delay.
Despite this lag, modern CGMs achieve MARD values of 7.9-14.2% — meaning readings are within 8-14% of a simultaneous blood glucose lab value on average. The Dexcom G7 leads the market at 8.2% MARD, followed by the FreeStyle Libre 3 Plus at 7.9% MARD in its pivotal trial. A 2023 Diabetes Care meta-analysis of 47 clinical trials (N = 12,847 participants) found that CGMs with MARD below 10% produced clinically equivalent insulin dosing decisions to fingerstick blood glucose meters in 96.8% of paired measurement comparisons. This evidence base is why the American Diabetes Association now endorses CGMs as a primary glucose monitoring tool rather than a supplement to fingerstick testing.
Accuracy varies by glucose range: most CGMs are most accurate in the 70-180 mg/dL range (normal to moderate hyperglycemia) and less accurate below 54 mg/dL (severe hypoglycemia, where MARD can exceed 20%) and above 350 mg/dL (severe hyperglycemia). This range-dependent accuracy is why CGM alerts for dangerously low glucose should always be confirmed with a fingerstick before treating with fast-acting carbohydrates — a recommendation endorsed by both Dexcom and Abbott in their product labeling. Sensor accuracy also improves over the first 12-24 hours of wear as the sensor stabilizes in the tissue, which is why many endocrinologists recommend inserting a new sensor 12 hours before removing the old one to minimize gaps in reliable data. For device-by-device MARD comparisons, see the CGM accuracy and MARD guide.
The Future of CGM Technology: What's Coming in 2027-2030
Several technology advances are in clinical development or early commercialization that will reshape the CGM market within 3-5 years. Non-invasive CGMs — devices that measure glucose through the skin without any needle or implant — are the most anticipated category. Companies including Biolinq (transdermal microelectrode array) and Know Labs (RF spectroscopy) are in FDA clinical trials, but neither has yet demonstrated MARD accuracy below 12% in peer-reviewed studies, compared to the 7.9-9.1% achieved by current electrochemical sensors. A truly viable non-invasive CGM must achieve sub-10% MARDto meet the clinical accuracy threshold established by the FDA's 2023 guidance document for integrated CGM systems.
Multi-analyte sensorsthat measure glucose plus ketones, lactate, or alcohol simultaneously are in development at both Dexcom and Abbott. Dexcom's patent filings (published 2024) describe a dual-enzyme sensor capable of reporting glucose and beta-hydroxybutyrate (the primary blood ketone) from a single subcutaneous filament — a breakthrough that would allow Type 1 diabetes patients to monitor for diabetic ketoacidosis without a separate blood ketone meter. Abbott's Lingo 2.0 roadmap, disclosed at the 2025 JP Morgan Healthcare Conference, includes lactate sensing for endurance athletes by 2028.
Extended-wear sensors lasting 30+ days would reduce the annual sensor count from 24 (current 15-day sensors) to 12 or fewer, cutting annual supply costs by up to 50%. Dexcom's G8 program targets a 21-day wear time with an anticipated launch in late 2027. AI-powered glucose prediction that forecasts blood sugar 60-120 minutes ahead using CGM data combined with meal photos and activity data is already available through services like January AI, though a 2025 Journal of Diabetes Science and Technology evaluation found prediction accuracy degrades significantly beyond 30-minute windows — mean prediction error was 11 mg/dL at 30 minutes but 34 mg/dL at 120 minutes. For current sensor specifications, see the CGM brands and manufacturers guide.
Deep Dives: CGM Technology Topics
CGM Sensor Technology: Electrochemical, Fluorescence, and Biosensor Systems
Read guide →
CGM Accuracy and MARD: Understanding the 8-14% Range Across Devices
Read guide →
Real-Time CGM vs Flash Monitoring: Continuous Data vs On-Demand Scans
Read guide →
CGM Calibration: Factory-Calibrated vs Fingerstick-Based Systems
Read guide →
CGM Data and Glucose Reports: AGP, Trends, and 288 Daily Readings
Read guide →
Time in Range: The 70% TIR Target and Why It Matters for A1C
Read guide →
CGM Alarms and Alerts: Predictive Urgent Low and 5 Alert Types
Read guide →
CGM Smartphone Apps: Bluetooth, Cloud Sync, and Real-Time Data Sharing
Read guide →
CGM and Insulin Pump Integration: 3 Closed-Loop Systems in 2026
Read guide →
When a CGM Isn't the Right Tool
Continuous glucose monitoring provides 288+ daily readings, but not every person needs that level of data density. For individuals with stable type 2 diabetes managed by oral medication alone, 4 to 10 daily fingerstick readings may provide sufficient insight at a fraction of the cost ($15-75/month vs $75-300/month for CGM). A quarterly lab A1C test — which reflects average glucose over 2 to 3 months — remains the diagnostic gold standard for long-term glucose control and costs just $25-50 per test. For a side-by-side comparison of all 7 blood sugar testing methods including accuracy, cost, and ideal use cases, see the blood sugar testing methods guide.
Related Resources
- Best CGMs Ranked for 2026 — Every continuous glucose monitor compared by MARD accuracy, wear time, and features.
- CGM Cost and Insurance Guide — Monthly pricing, Medicare coverage, OTC affordability, and how to reduce out-of-pocket costs.
- 5 CGM Services with App Integration and Coaching — Nutrisense, Levels, Signos, Veri, and January AI — pricing, data features, and support.
- CGM Brands and Manufacturers — Full specifications for every device from Dexcom, Abbott, Medtronic, and Senseonics.