What Causes Blood Sugar Spikes: 9 Triggers and Prevention Strategies
9 factors cause blood sugar spikes — refined carbs, stress, poor sleep, dehydration, illness, medications, caffeine, dawn phenomenon, and inactivity. Prevention strategies with CGM data for each trigger.
What Is a Blood Sugar Spike?
A blood sugar spike is a rapid rise in blood glucose concentration — typically exceeding 30 to 50 mg/dL above baseline within 30 to 60 minutes. In clinical terms, a spike above 180 mg/dL is classified as hyperglycemia by the American Diabetes Association, while the International Diabetes Federation defines a clinically significant postmeal spike as any excursion above 160 mg/dL. For non-diabetic adults, blood sugar rarely exceeds 140 mg/dL after meals under normal conditions. The speed and height of a glucose spike matters because rapid glucose fluctuations — quantified as the Mean Amplitude of Glycemic Excursions (MAGE) — cause greater oxidative stress and endothelial damage than sustained moderate hyperglycemia at the same average glucose level. A 2006 study by Monnier et al. in JAMA demonstrated that glucose variability (repeated spikes and crashes) activated more markers of oxidative stress than stable high glucose. CGMs measure glucose every 1 to 5 minutes, making spikes visible as sharp upward slopes on the trend line that can be correlated with specific foods, activities, and events in real time.
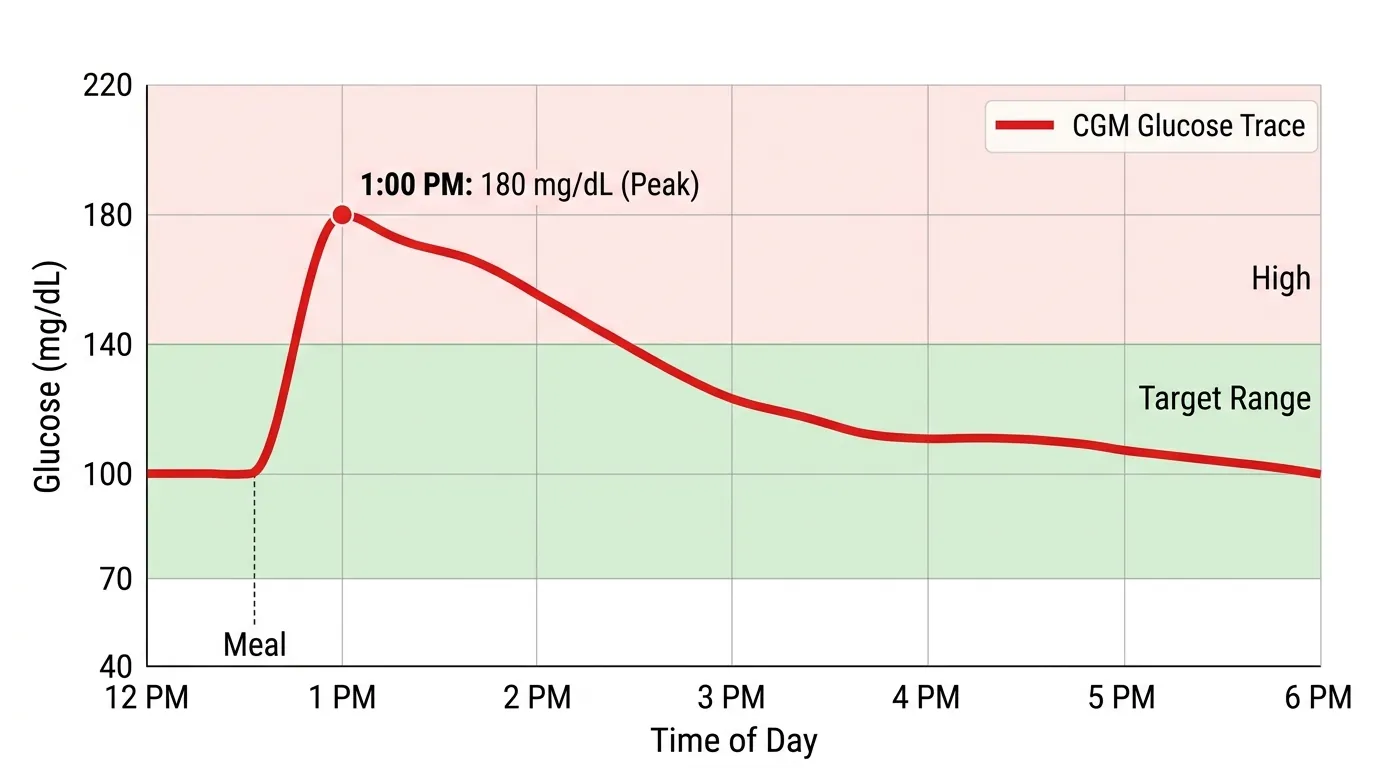
Trigger 1: Refined Carbohydrates and Added Sugars
Refined carbohydrates are the most potent and most common trigger for blood sugar spikes. Foods made from white flour, white rice, and added sugars are rapidly digested into glucose because their fiber has been stripped during processing. A single 12-ounce can of regular soda contains 39 grams of sugar and raises blood sugar by 60 to 100 mg/dL within 20 minutes. Two slices of white bread (26 grams of refined carbs) produce a peak glucose rise of 40 to 70 mg/dL in 30 to 45 minutes. The glycemic index (GI) quantifies this effect on a 0-100 scale: white bread scores 75, white rice scores 73, and pure glucose is the reference at 100. Foods with a GI below 55 (most vegetables, legumes, whole grains) produce flatter, lower glucose curves. The glycemic load (GL) combines GI with carbohydrate quantity per serving, providing a more practical prediction of glucose impact. Prevention strategies include swapping refined grains for whole grains (reduces spike by 20-30%), adding vinegar to meals (1 tablespoon of apple cider vinegar before a meal reduces postmeal glucose by 20-30% per a 2004 study by Johnston et al. in Diabetes Care), and pairing carbohydrates with protein, fat, and fiber.
Trigger 2: Physical and Psychological Stress
Stress raises blood sugar through the hypothalamic-pituitary-adrenal (HPA) axis, which releases cortisol and adrenaline. These hormones signal the liver to release stored glucose and simultaneously reduce insulin sensitivity in muscle and fat cells — a survival mechanism that ensures the brain and muscles have fuel during a perceived threat. Acute stress (a work deadline, an argument, a traffic jam) can raise blood sugar by 30 to 50 mg/dL within 15 to 30 minutes even without eating. Chronic stress maintains cortisol at elevated levels, raising fasting glucose by 10 to 20 mg/dL over weeks to months. A 2019 study at Ohio State University (Hackett et al., Psychoneuroendocrinology) tracked 100 non-diabetic office workers with CGMs and cortisol monitors for 5 days and found that self-reported stress episodes preceded blood sugar rises of 25 to 45 mg/dL on average, with the glucose effect lasting 1 to 3 hours after the stressor resolved. Prevention strategies include regular aerobic exercise (lowers baseline cortisol by 15-20%), diaphragmatic breathing (activates the parasympathetic nervous system, reducing cortisol release), and consistent sleep schedules. CGM data helps users identify stress-driven glucose patterns by correlating spikes with non-meal events.
Triggers 3-5: Sleep, Dehydration, and Illness
Poor sleep, dehydration, and illness each independently raise blood sugar through distinct mechanisms. Sleep deprivation (fewer than 6 hours) reduces insulin sensitivity by 25 to 40% and increases cortisol by 37%, as demonstrated in a landmark study by Spiegel et al. at the University of Chicago (The Lancet, 1999). The effect is dose-dependent: each hour of sleep lost below 7 hours raises fasting glucose by approximately 5 mg/dL and increases postmeal glucose peaks by 10 to 20 mg/dL the following day. Dehydration concentrates blood glucose — losing 2% of body weight in water increases blood sugar readings by 10 to 15 mg/dL simply through hemoconcentration, independent of any metabolic change. The American College of Sports Medicine recommends maintaining urine color at pale yellow (specific gravity below 1.020) for accurate glucose readings. Illness triggers the release of counter-regulatory hormones (cortisol, glucagon, growth hormone, catecholamines) that collectively raise blood sugar by 40 to 100+ mg/dL depending on severity. Even a mild upper respiratory infection raises fasting glucose by 20 to 40 mg/dL. During illness, the ADA recommends checking blood sugar every 2 to 4 hours — a task that CGMs automate, providing critical overnight monitoring when illness-related hyperglycemia is most dangerous.
Triggers 6-7: Medications and Caffeine
Multiple medication classes raise blood sugar as a side effect, sometimes dramatically. Corticosteroids (prednisone, dexamethasone) are the most potent — a single 40mg dose of prednisone raises blood sugar by 40 to 100+ mg/dL within 4 to 8 hours by increasing hepatic glucose output and reducing peripheral insulin sensitivity. This effect persists for the duration of steroid therapy and for 24 to 48 hours after the last dose. Thiazide diuretics raise fasting glucose by 5 to 15 mg/dL through potassium depletion and direct beta-cell inhibition. Beta-blockers increase fasting glucose by 5 to 10 mg/dL and mask the symptoms of hypoglycemia (tremor, rapid heart rate), making them particularly problematic for people on insulin. Atypical antipsychotics (olanzapine, clozapine) increase diabetes risk by 30 to 60% through weight gain and direct effects on insulin signaling. Caffeine in doses above 200 mg (approximately 2 cups of coffee) raises blood sugar by 8 to 15 mg/dL by stimulating adrenaline release and temporarily reducing insulin sensitivity. A 2008 study in Diabetes Care found that 250 mg of caffeine increased postmeal glucose peaks by an average of 21% in people with type 2 diabetes. CGM data is invaluable for isolating medication effects on glucose because the continuous recording captures the time-locked glucose rise that follows a specific dose.
Triggers 8-9: Dawn Phenomenon and Physical Inactivity
The dawn phenomenon is an early-morning blood sugar rise caused by the natural circadian release of cortisol, growth hormone, and glucagon between 3 AM and 8 AM. These hormones signal the liver to release glucose to fuel the transition from sleep to wakefulness. The dawn phenomenon affects approximately 50% of people with diabetes and raises fasting blood sugar by 20 to 60 mg/dL compared to the overnight nadir. It is distinct from the Somogyi effect, which is a rebound hyperglycemia following nocturnal hypoglycemia — the Somogyi effect involves a glucose dip below 60 mg/dL followed by a counter-regulatory surge, while the dawn phenomenon shows a steady rise without any preceding low. Only a CGM can reliably distinguish between these two mechanisms. Physical inactivity is the chronic trigger that amplifies all others. Skeletal muscle is the largest glucose disposal organ in the body, absorbing 80% of postmeal glucose. When muscles are inactive, their glucose uptake decreases by 35 to 40% within 24 hours (Hamilton et al., Diabetes, 2007). A 10-minute walk after meals lowers the postmeal glucose peak by 22% (Reynolds et al., Sports Medicine, 2016). Regular exercise — 150 minutes per week of moderate activity — reduces fasting glucose by 10 to 20 mg/dL and postmeal peaks by 15 to 30 mg/dL within 2 weeks by increasing GLUT4 transporter expression in muscle cells.