Senseonics received FDA clearance for the Eversense 365 in February 2025, making it the first continuous glucose monitor that can remain implanted under the skin for a full 365 days. The device represents a fundamentally different approach to glucose monitoring—instead of a disposable sensor replaced every 7-15 days, Eversense 365 uses a tiny implantable fluorescence-based sensor that requires just one minor outpatient procedure per year.
How Fluorescence-Based Sensing Works
Unlike conventional CGMs that use electrochemical (enzyme-electrode) technology, the Eversense sensor measures glucose through fluorescence. The implant is a small cylindrical sensor (approximately 3.5 mm in diameter and 18.3 mm long) coated with a glucose-responsive fluorescent polymer.
Here is the sensing process: an external transmitter worn on the skin above the implant shines near-infrared light through the tissue. The light excites the fluorescent coating on the sensor, and the amount of fluorescence emitted changes proportionally to the glucose concentration in the surrounding interstitial fluid. The transmitter measures the fluorescent response, calculates the glucose value, and sends it to a smartphone app via Bluetooth.
The fluorescent polymer is inherently more stable over time than electrochemical enzyme layers, which is why Eversense sensors can last 365 days while traditional sensors degrade after 7-15 days. Clinical trials demonstrated an overall MARD of 8.5 percent for the Eversense 365, competitive with the best disposable CGMs on the market.
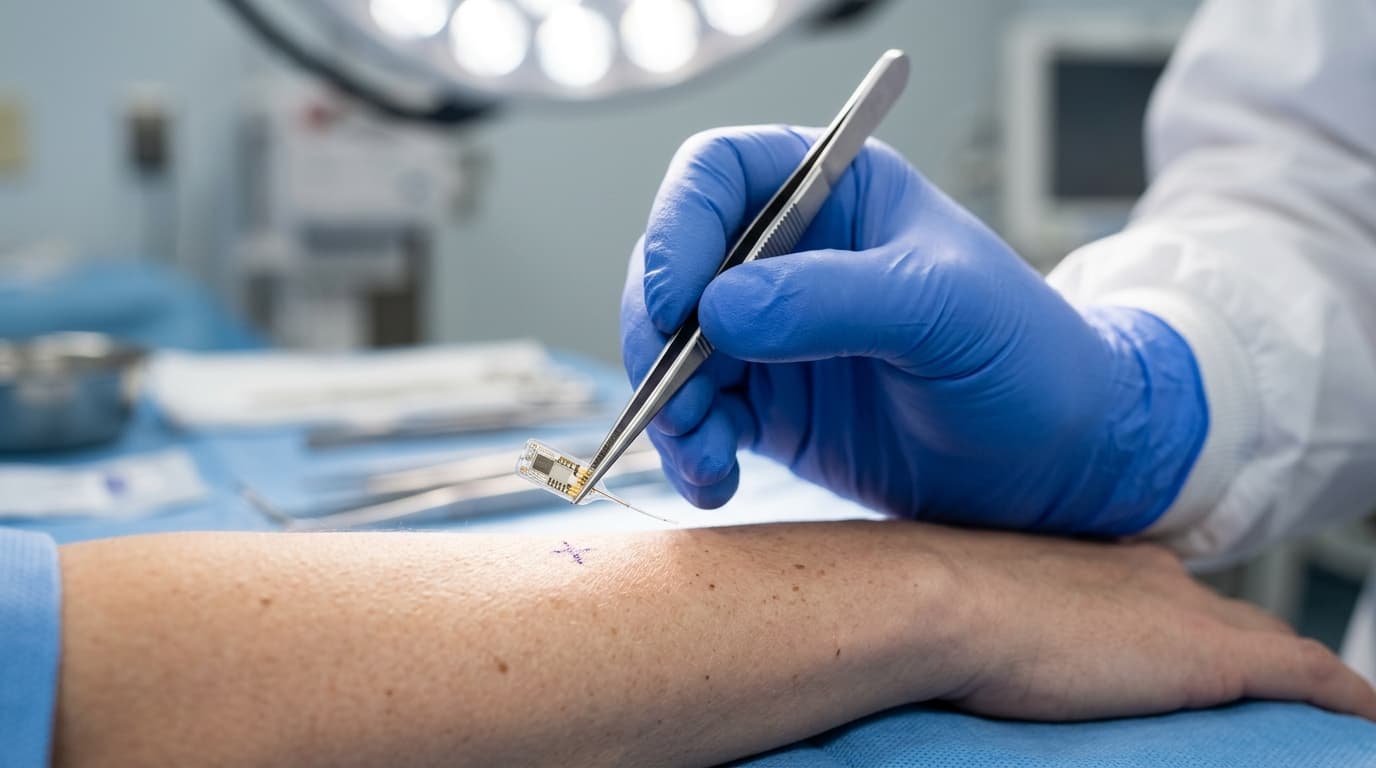
The Implant Procedure
The Eversense sensor is inserted by a trained healthcare provider in a brief outpatient procedure that takes approximately 5-10 minutes. The provider makes a small incision (less than 1 cm) on the upper arm, places the sensor into the subcutaneous tissue, and closes the site with adhesive strips or a single stitch. Local anesthesia is used; no sedation is required.
After implantation, a 24-hour warmup period is required before the sensor begins reporting glucose values. Removal at the end of 365 days follows a similar process: a small incision, sensor extraction, and wound closure. A new sensor can be implanted in the same visit if the patient chooses to continue.
The most common side effects from clinical trials were mild: insertion site discomfort (experienced by 7 percent of participants), minor bruising, and temporary skin irritation from the external transmitter adhesive. Serious adverse events related to the implant were rare, occurring in fewer than 1 percent of participants.
The External Transmitter
The Eversense system requires a rechargeable external transmitter worn over the implant site on the upper arm. The transmitter attaches to the skin using a silicone adhesive and must be worn continuously for glucose readings. It is water-resistant but should be removed for charging (approximately 15 minutes daily) and for showering if preferred.
One unique feature of the Eversense transmitter is on-body vibration alerts. Unlike other CGMs that rely solely on smartphone notifications, the Eversense transmitter vibrates directly on the arm when glucose goes above or below the set thresholds. This provides a tactile alert that works even if the phone is in another room or on silent mode.
Who the Eversense 365 Is Designed For
Senseonics positions the Eversense 365 for people with diabetes who are frustrated by the inconvenience of frequent sensor changes and adhesive-related skin problems. The ideal candidate is a person with type 1 or type 2 diabetes (on intensive insulin therapy) who:
- Experiences adhesive allergies or skin irritation from disposable CGM sensors - Wants to minimize the visible presence of diabetes technology on their body - Values the reliability of a sensor that does not need to be replaced every 1-2 weeks - Is comfortable with a minor outpatient procedure twice per year (insertion and removal)
Cost Considerations
The Eversense 365 sensor costs approximately $3,000-$4,000 per year for the sensor and transmitter. However, because the sensor lasts 365 days, the annual cost is comparable to 12 months of disposable CGM sensors (which run $1,200-$3,600 per year depending on the brand and insurance coverage).
Insurance coverage for Eversense varies by plan but is increasingly available. Medicare covers the Eversense system under Part B for qualifying beneficiaries, and most major commercial insurers include it on their formularies. The insertion and removal procedures are covered as outpatient surgical services.
The Eversense 365 will not appeal to everyone—many CGM users prefer the simplicity of sticking on a new sensor without a medical appointment. But for a significant subset of the diabetes community, the prospect of only 2 clinic visits per year and zero sensor changes in between represents a meaningful quality-of-life improvement.